ACH 101: Introduction to Inorganic Chemistry
| Institution | UNIVERSITY |
| Course | BIOCHEMISTRY |
| Year | 1st Year |
| Semester | Unknown |
| Posted By | Brian Mike |
| File Type | |
| Pages | 23 Pages |
| File Size | 1.52 MB |
| Views | 3611 |
| Downloads | 0 |
| Price: |
Buy Now
|
Description
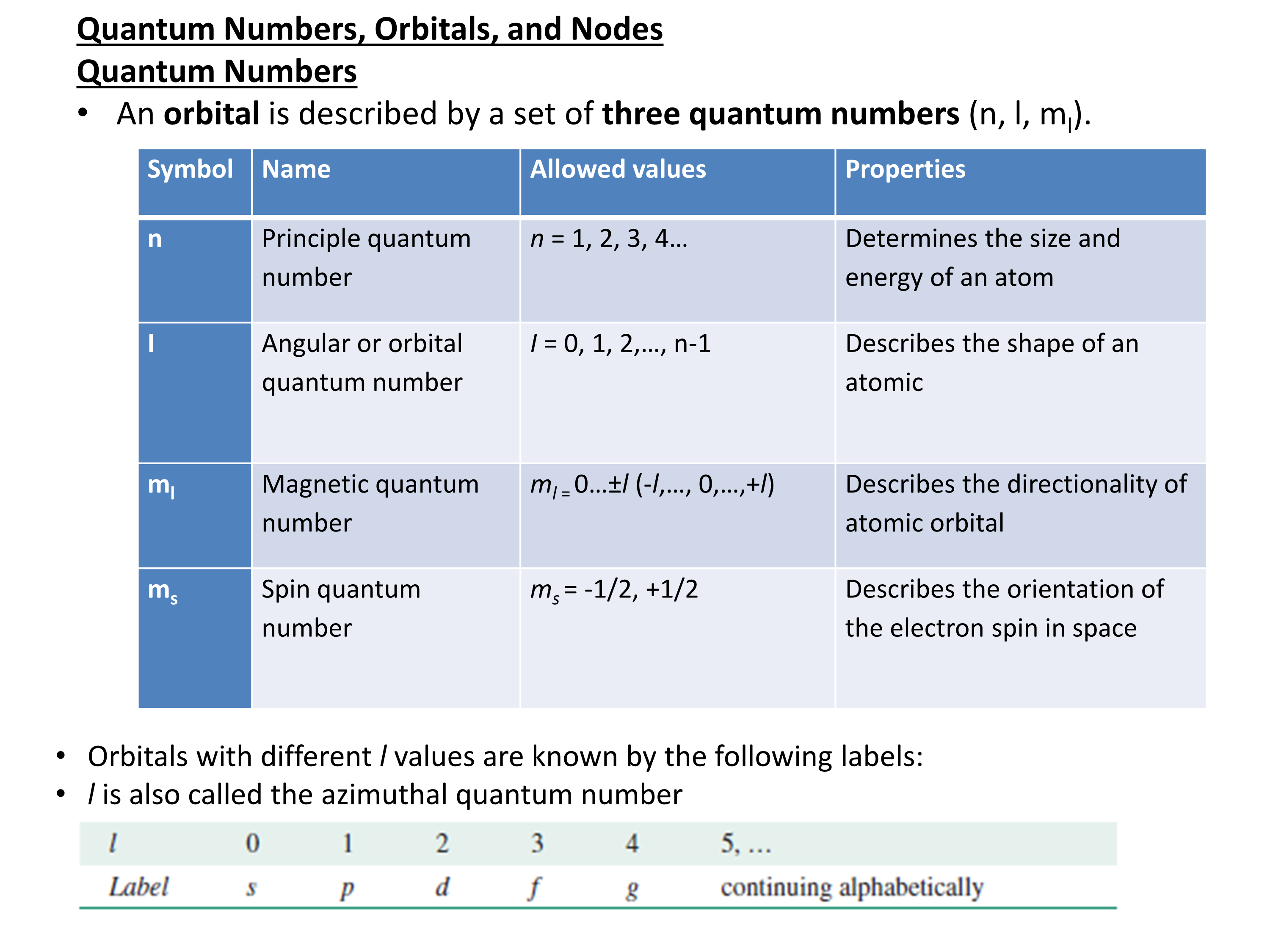
The electronic configuration of cations is assigned by removing electrons first in the outermost p orbital, followed by the s orbital and finally the d orbitals (if any more electrons need to be removed).
Example:
calcium (Z=20), 1s22s22p63s23p64s2; Ca2+: EC is 1s22s22p63s23p6
chlorine (Z=17), EC is 1s22s22p63s23p5 Cl- : 1s22s22p63s23p6.
The ns electrons are always lost before the (n-1)d when forming cations for transition metals. For example,
Zn: [Ar]4s23d10 Zn+2: [Ar]3d10
Below is the document preview.
History form 2 exams paper 1 and 2 (with marking schemes)
Trending!
This entails form 2 exams for term two.Besides,it has both paper 1 and 2.Therefore it is very useful for your students
6862 Views
10 Downloads
40.47 KB
Elegant business card template
Trending!
Photoshop psd elegant business card template free
7372 Views
0 Downloads
2.62 MB
Awesome insiring web design template
Trending!
Free awesome responsive bootstrap web design template
3883 Views
0 Downloads
2.8 MB
BIOCHEMISTRY
Polyurethanes are prepared from diisocyanates
and diols. Urethane (also called a carbamate) is a
functional group that contains both an ester and an
amide at a single carbonyl group.
Nucleophilic Addition Reaction: Example 1:
Sevin is a highly effective, biodegradable insecticide. It
affects the nervous systems of insects by blocking
access to the neurotransmitter acetylcholine.
57 Pages
775 Views
0 Downloads
1.68 MB
BIOCHEMISTRY
PETROLEUM CHEMISTRY
Carbon is so Important in the Life Cycle. It has
numerous ways of bonding with many other
elements, particularly oxygen and hydrogen.
It can form both “organic” and “inorganic”
compounds. Organic compounds are considered
unstable in the biosphere because they are in the
reduced state.
Whereas Inorganic compounds, principally
calcite (CaCO3
) and dolomite (CaMg(CO3
)2
), are
stable because they are in the oxidized state.
Carbon is present in most substances that are
vital for the development of life (called
“biomolecules”): Proteins, lipids, sacharides, etc
76 Pages
714 Views
0 Downloads
1.86 MB
BIOCHEMISTRY
Alkyl halides are organic molecules containing a halogen
atom bonded to an sp3 hybridized carbon atom.
• Alkyl halides are classified as primary (1°), secondary (2°),
or tertiary (3°), depending on the number of carbons bonded
to the carbon with the halogen atom.
• The halogen atom in halides is often denoted by the symbol
“X”.
53 Pages
721 Views
0 Downloads
2.07 MB
BIOCHEMISTRY
Natural Gas
Natural gas is a gaseous fossil fuel consisting
primarily of methane but including significant
quantities of ethane, butane, propane, carbon
dioxide, nitrogen, helium and hydrogen sulfide.
Obtained from oil fields and natural gas fields,
and in coal beds.
Methane-rich gases are produced by the
anaerobic decay of non-fossil organic material, ie
biogas.
Total world production of natural gas in 1986
was 100 trillion m3
.
It is used as feed stock as well as fuel. It is
preferred due to its high Calorific Value
52 Pages
679 Views
0 Downloads
1.17 MB
Calculus 1 First Year
Trending!
Excelling in Calculus is the key
81 Pages
3635 Views
2 Downloads
841.91 KB